Product focus
Product focus
IRIDEX Cyclo G6™
MicroPulse Technology™
Powered by the NEW Cyclo G6™ Glaucoma Laser System
The Cyclo G6 laser system is dedicated specifically to treat patients diagnosed with a range of glaucoma disease states and features the company's proprietary Micropulse® tissue-sparing technology and family of single use probes that connect to an intuitive, user-friendly laser console.
Some studies of the Cyclo G6 have shown a 30-40% reduction in intraocular pressure (IOP) so this is a very promising therapy for our patients. In addition, the ability to reduce IOP without incisions nor using implants in a quick procedure is very attractive, as is the repeatability, potentially extending the time before more invasive measures are required." - Keith Barton, Consultant Ophthalmologist, Glaucoma service, Moorfields Eye Hospital.
IRIDEX Cyclo G6™
The Cyclo G6 laser system is dedicated specifically to treat patients diagnosed with a range of glaucoma disease states and features Micropulse® tissue-sparing technology.
Innovative Cyclophotocoagulation with MicroPulse Technology Powered by the NEW CYCLO G6™ Glaucoma Laser System.
View product details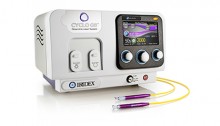

Products
Products by type
Diagnostic ImagingOphthalmic Diagnostic Equipment - Aiding Social DistancingOphthalmic LensesSlit Lamps and Ophthalmic LensesTonometry and Visual FieldsSight Testing and RefractionCornealConsultation FurnitureUltrasoundSurgical BedsLasers and ProbesSurgicalConsumablesProducts
Products by brand
Arc Optical LtdBELKIN VisionBon OpticBovieChongqing Sunkingdom MedicalCorza OphthalmologyESI Inc.EvansInvotecIridexKeelerMeccanotticaMediWorksMurrayNeoLightOculusOptotek MedicalPlusoptixReichertRodenstock InstrumentsSurgistarTakagiUFSKVELAVisionixVolkZiemerService
Engineering support Clinic design serviceSpecial Offers
Current offersProduct Focus
IRIDEX Cyclo G6™Reichert® Tono-Vera®BELKIN Vision Eagle™MicroPulse®Takagi 700GL LEDUFSK 500XLEVELA ‘Move+’Corza Medical Hands-FreeNeoLight Phoenix ICONPolicies & Certificates
ISO Certificate WEEE Policy GDPR Policy GDPR Auth form Social Responsibility Document Modern Slavery Statement Sustainability ReportCarleton Optical
Pattisson House
Addison Road, Chesham
Buckinghamshire HP5 2BD
UK
Email:
carleton@carletonltd.comSales:
01494 775 811
 Subscribe via RSS
Subscribe via RSS
 Join us on LinkedIn
Join us on LinkedIn
 Follow us on Twitter
Follow us on Twitter
 Find us on Facebook
Find us on Facebook




























